Principle Investigators:
Zoi-Dorothea Pana, Andreas H. Groll, Thomas Lehrnbecher, Sarina Butzer, Luise Martin, Daniel Ebrahimi-Fakhari, Oliver A. Cornely, Danila Seidel

Overview:
The Pediatric Isavuconazole Registry (Ped-IR) is a multinational retrospective study conducted within the FungiScope® registry. This study aims to improve understanding of how isavuconazole is used in real-world clinical practice to treat invasive mold infections in children.
Inclusion criteria:
- Pediatric patients (< 18 years of age at the time of isavuconazole administration)
- Receiving isavuconazole for treatment of proven/probable/possible invasive mold infection, i.e., aspergillosis, mucormycosis and others (for more details, please see study protocol below)
- Start of isavuconazole treatment from January 2024 to December 2025
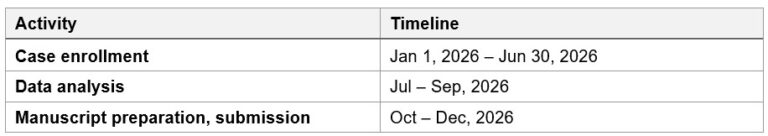
Timeline for Ped-IR:
Further information and details on Ped-IR for download:
For participation, please contact the FungiScope® Study Team: fungiscope@uk-koeln.de.
